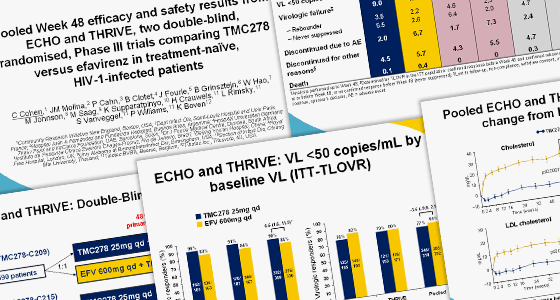
Images from presentation by Dr Cal Cohen of Community Research Initiative New England (www.crine.org)
Rilpivirine (TMC278) is as effective as efavirenz (Sustiva or Stocrin) when used in combination antiretroviral therapy by people starting treatment, pooled results from two studies presented to the Vienna conference show.
The safety and effectiveness of rilpivirine, an experimental non-nucleoside reverse transcriptase inhibitor (NNRTI), made by Tibotec, was compared to that of efavirenz in the ECHO and THRIVE studies. These trials involved 1400 patients starting HIV treatment for the first time.
After 48 weeks of the trial, the proportion of people in each arm with an undetectable viral load was almost identical (84.3% in the rilpivirine arm, 82.3% in the efavirenz arm). This showed that rilpivirine was ‘non-inferior’ to efavirenz.
Patients who took efavirenz were more likely to stop treatment than those taking rilpivirine (6.7 vs 2%). Those taking efavirenz were about three times more likely to report side-effects such as dizziness and vivid dreams.
Resistance patterns differed amongst those efavirenz- and rilpivirine-treated patients whose viral load rebounded. Rilpivirine-treated patients tended to develop the E138K mutation that causes resistance to the second-line NNRTI etravirine (Intelence). Half of those who experienced treatment failure on rilpivirine developed resistance to the drug and, of them, 90% were resistant to etravirine too.
It is expected rilpivirine will be submitted for a US licence very soon, and it is likely that it will be combined into a single, once-daily pill with Gilead’s Truvada (tenofovir and FTC).